Dry Eye Disease
Introduction
Dry eye disease (DED) is a common issue that affects hundreds of millions of people worldwide, making it one of the top reasons for visits to eye care practitioners.1 Depending on location in the world, its prevalence can be anywhere from 8.7% to 64%.2
Estimating DED incidence is challenging, as symptom reports can fluctuate, especially in mild cases. Claims data may also be affected by reimbursement changes or new therapies introduced during a study3.
For some, dry eye is just an occasional annoyance. But for others, moderate to severe DED can bring significant pain, make daily activities tough, lower energy levels, impact overall health, and even lead to depression. Quality of life (QoL) is influenced by various factors, especially when it comes to mental health and its social and economic repercussions. Interestingly, when it comes to vision-related QoL, DED has quite an impact, showing similar or even greater effects compared to conditions like macular degeneration, glaucoma, retinal detachment, and allergic conjunctivitis4. Research has found an interesting connection between DED and feelings of depression, anxiety, and mood swings5. One of the main reasons behind this is that many patients dealing with DED aren’t aware that there’s help available, so they don’t go looking for treatment. Recent studies even suggest that not getting diagnosed is associated with worse mental health-related quality of life for those with severe DED4.
If severe dry eyes are left untreated, it can cause serious problems such as eye infections, inflammation, corneal abrasions, corneal ulcers, and even vision loss. So, it’s crucial for patients to address the symptoms early and seek appropriate treatment.
In 2025, the TFOS Dry Eye Workshop (DEWS III) updated the definition of dry eye:
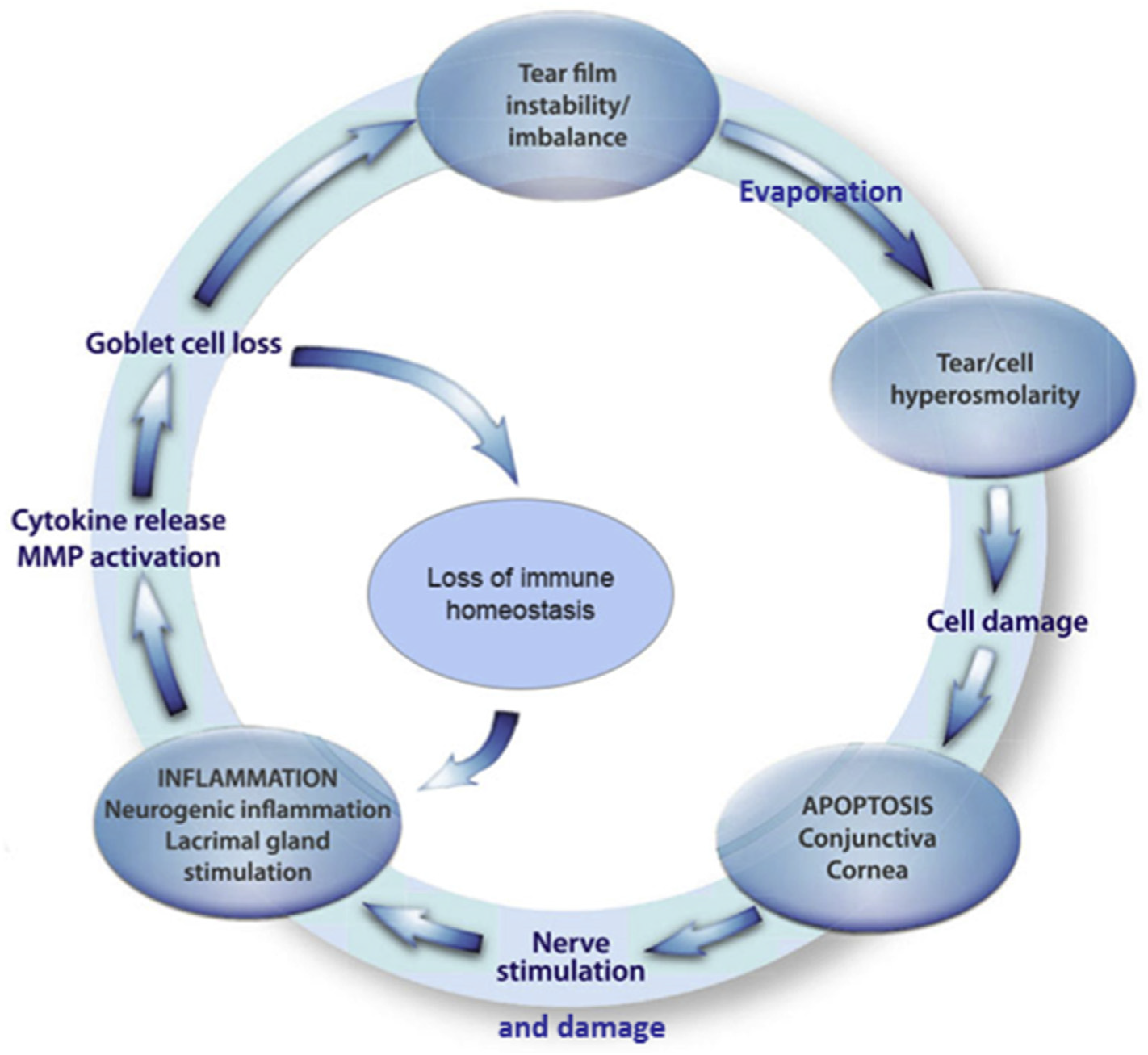
Dry eye is a multifactorial, symptomatic disease characterized by a loss of homeostasis of the tear film and/or ocular surface, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities are etiological factors6.
For more details on DEWSIII, click here.
The result of this is that dry eye is now recognised as a symptom-led, multifaceted disease where the tear film and/or the ocular surface lose their balance. The condition comes with various eye symptoms and involves tear film instability, high tear film saltiness/ hyperosmolarity, inflammation and damage to the eye surface, and issues with eye nerve sensations. This is often referred to as the self-perpetuating vicious cycle of DED8.
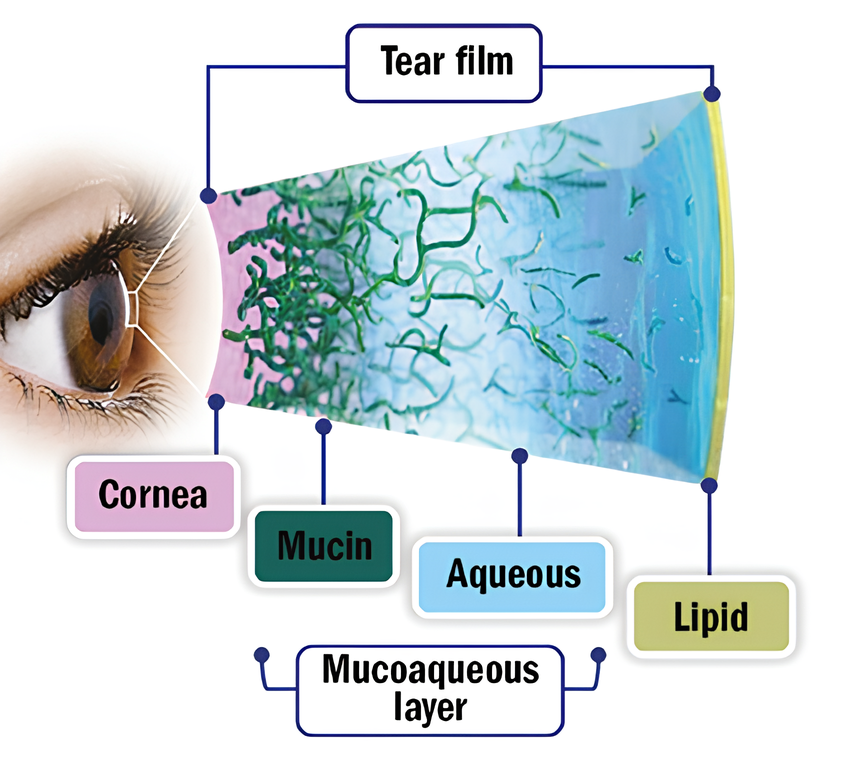
To really grasp the mechanisms behind dry eye disease, it’s key to understand the tear film’s structure and function. The tear film is a complex layer that covers the eye’s surface. It plays several crucial roles: protecting, nourishing, and lubricating the eye, as well as forming the first refractive surface of the visual system. Although it’s a bit of an oversimplification, the tear film is still often described using the classic three-layer model. However, more recently, it is encouraged to think of it as more of a mucoaqueous continuum. This means there’s a higher concentration of mucins closer to the eye’s surface and more aqueous fluid moving towards the lipid layer3.
Traditionally, the function of the different layers are as follows:
- The innermost mucin layer helps the tears stick to the surface of the eye.
- The middle aqueous layer keeps the eye nourished and protected.
- The outermost lipid layer keeps everything lubricated and stops the tears from evaporating.
Historically, DED was subclassified into two main types: aqueous deficient dry eye (ADDE) and evaporative dry eye (EDE), with EDE being the most common. It is important to keep in mind that these types often overlap and can occur together. In fact, around 80% of all DED cases are a mix of both1.
TFOS DEWS III now suggests that DED is subclassified according to the etiological drivers, tear film deficiencies, eyelid anomalies, ocular surface abnormalities and associated systemic conditions6.
Tear film deficiencies: Lipid, aqueous and/or mucin/glycocalyx deficiencies.
Eyelid anomalies: Blink/lid closure and lid margin disease
Ocular surface abnormalities: anatomical misalignment, neural dysfunction, ocular surface cell damage/disruption, primary inflammation/oxidative stress
Associated systemic conditions: Autoimmune conditions, hormonal imbalance and metabolic disease
Identification of the relevant driver of disease allows for targeted management. Due to the multifactorial nature of DED, it is common for patients to have multiple drivers contributing towards the disease.
Clinical Evaluation
Risk Factors
Risk Factors for Dry Eye as reported in the TFOS DEWS III Digest Epidemiology section are as follows:3
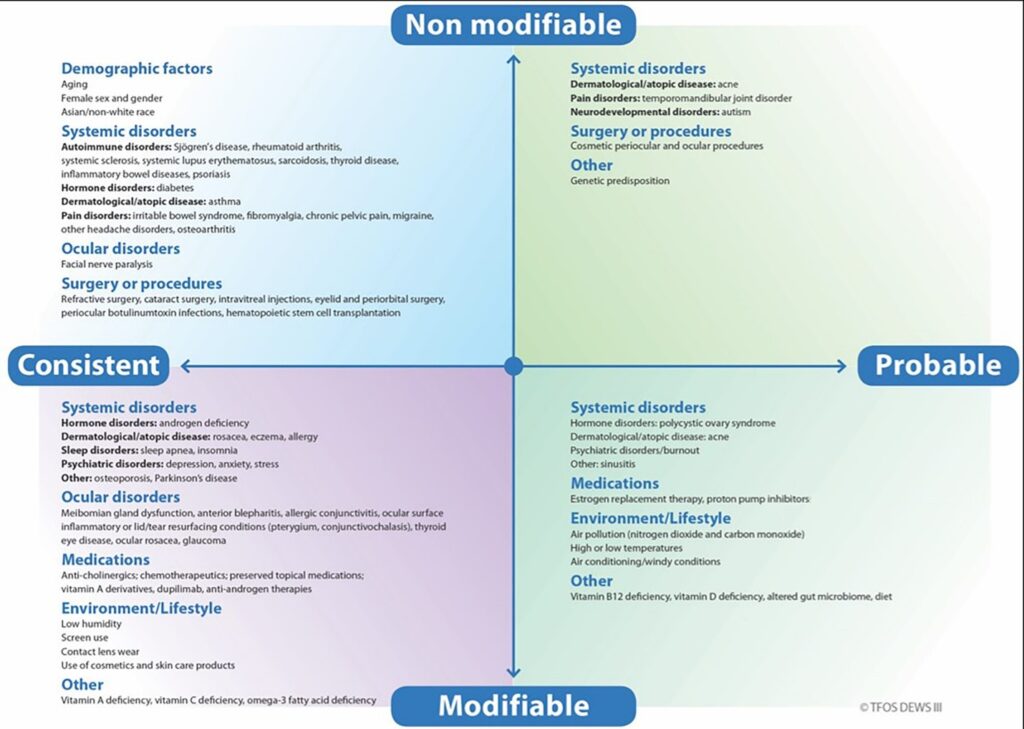
Frequently described risk factors with inconclusive evidence include: smoking, caffeine intake, alcohol intake, water intake, food restriction, mediterranean diet (possible positive effect), menopause, air pollution, oral contraceptives.
DED is a complex condition with many contributing factors, making it challenging to diagnose and manage. Understanding patients, including their eye health history and lifestyle, helps manage DED more effectively. DED usually arises from a mix of factors which can include:
Blepharitis (Click for more information)
Meibomian Gland Dysfunction (Click for more information)
Hormonal dry eye: Androgens play a crucial role in regulating the lipid and aqueous layers of the tear film3. Low androgen levels have consistently been linked to the development of DED9.
Paediatric dry eye: One study found that dry eye in children is linked to using visual display units and smartphones, with 11.03% of children affected by DED10. This is thought to be due to a reduced blink rate and an increase in conditions like blepharitis and meibomian gland dysfunction, making dry eye worse.
Diabetes and dry eye: Diabetes impacts the blood supply to the lacrimal gland, leading to less moisture release. It also reduces corneal sensitivity. This means that the eye can’t detect dryness and properly regulate tears, resulting in dry eye. The better a patient’s diabetes is controlled, the less severe their dry eye tends to be11.
Cosmetics and dry eye: Using eye cosmetics can lead to corneal nerve irritation, make the tear film hyperosmolar, cause meibomian gland dysfunction, and make the tear film unstable. This, in turn, can lead to dry eye symptoms as the makeup migrates onto the surface of the eye12 (Laura M. Periman, 2019).
Medications and Dry eye: Medications play a big role in causing dry eye, specifically iatrogenic dry eye3 (which is when medical treatment itself causes the problem). Click here to find out which medications can lead to DED. [Link to iatrogenic dry eye leaflet]
Refractive surgery and dry eye: Surgical manipulation of the cornea can disrupt the balance of the corneal nerve plexus (Eye Centre of Texas, 2022), leading to temporary dysfunction of the lacrimal functioning unit (LFU).
Cataract surgery and dry eye: Cataract surgery often leads to DED3 due to corneal nerve injury but also damage from the toxic components in eye drops. In fact, 100% of patients have shown abnormal tear break-up time (TBUT) and DED symptoms 12 weeks after surgery13.
Signs
Signs of dry eye include can include the following 14
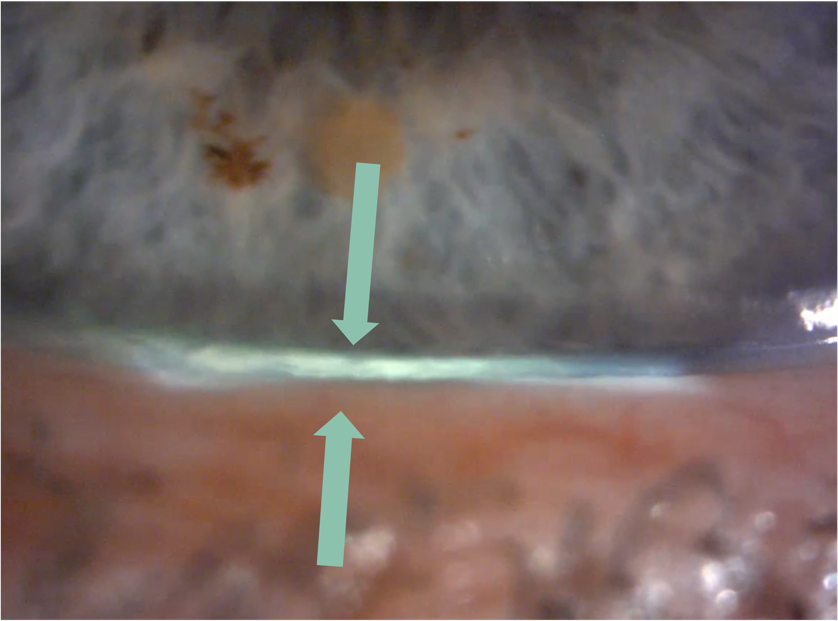
- Reduced tear meniscus at inferior lid margin (following the instillation of fluorescein, normal meniscus is not less than 0.2 mm in height)
- Raised tear osmolarity (308 mOsm/l is the most sensitive threshold to distinguish normal from mild/moderate DED, while 315 mOsm/l is the most specific cut-off)
- NITBUT (Non-invasive tear break up time) approx. <10 seconds
- Fluorescein break-up time (FBUT) <5 seconds
- Schirmer test (without anaesthesia) ≤ 5mm in 5 minutes; may be helpful in the diagnosis of Sjögren’s Syndrome, but of limited value in non-Sjögren’s DED
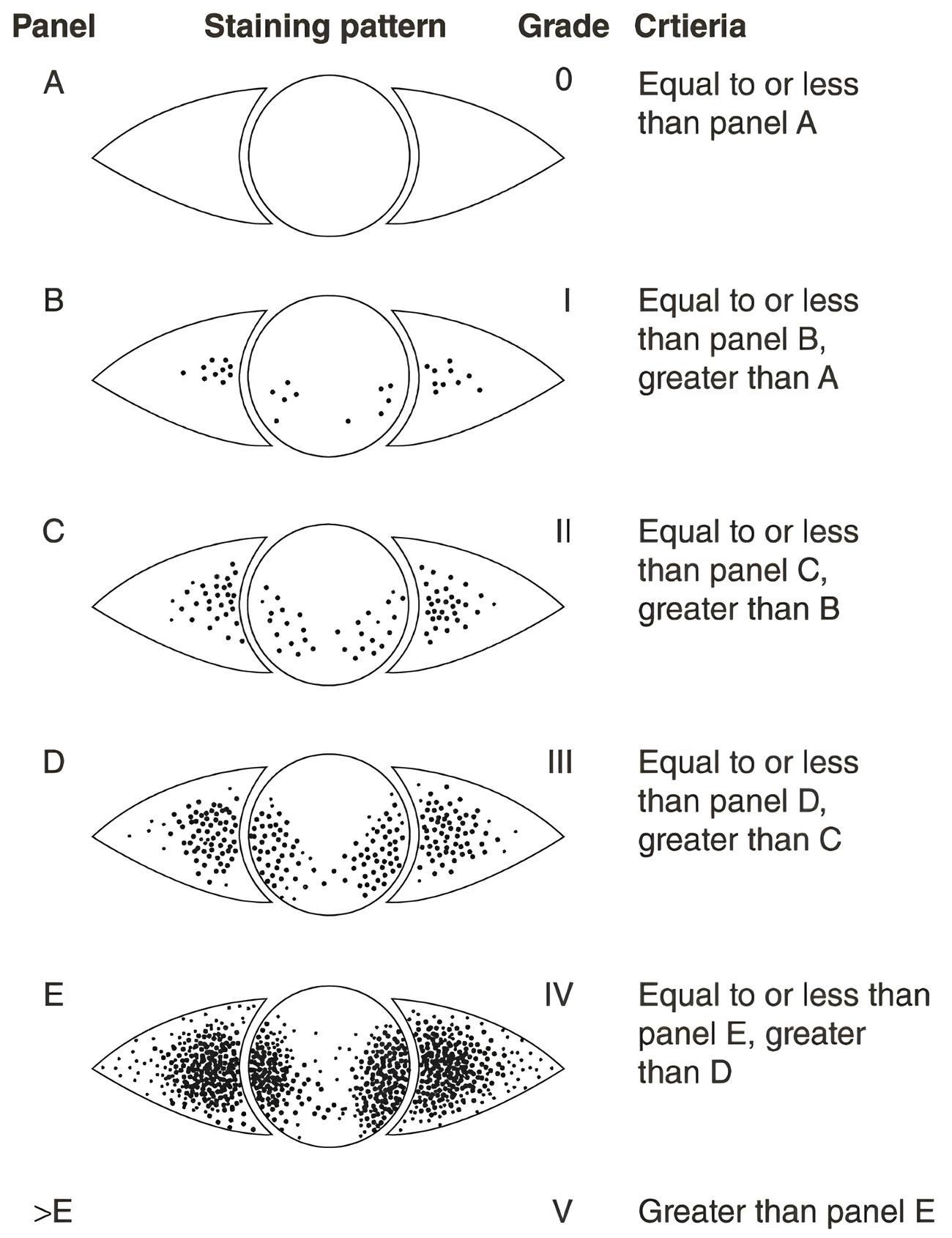
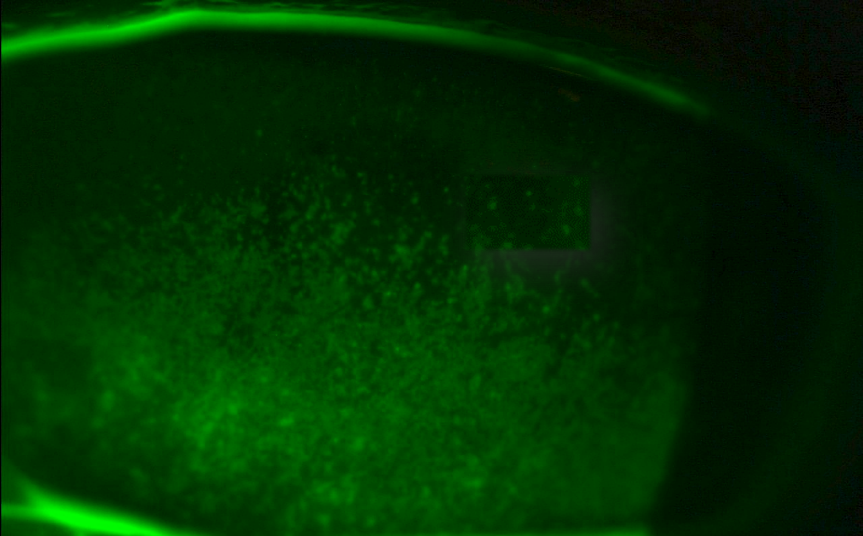
- Punctate epithelial erosions in exposed area of cornea and bulbar conjunctiva (especially in inferior third of palpebral aperture). Stain with vital dye(s) as available. Various grading systems are available (e.g. Oxford staining score)
- Lid wiper epitheliopathy
- Increased mucus strands and other tear film debris
- Filaments (adherent comma-shaped mucus strands)
- Mucus plaques
- Dellen
- Thinning and (very rarely) perforation
- Reduced corneal sensitivity
Symptoms
Symptoms of DED can include the following:
- Foreign body sensation
- Itching/irritation
- Tearing
- Redness
- Blurring/Fluctuation of vision
- Usually, bilateral
- Contact lens intolerance
- Excess tearing/watery eyes
TFOS DEWS III recommends the use of the OSDI Questionnaire to assess patient symptom severity in clinic6.
Research has shown that there isn’t a straightforward connection between the signs and symptoms of DED.15 For instance, Nichols et al., in a 2004 study examining the relationship between signs and symptoms in dry eye patients, concluded that “these findings suggest a weak connection between dry eye assessments and symptoms, posing a challenge in both clinical research and practice”.
While it’s true that the relationship between signs and symptoms isn’t always straightforward in cases of DED, it’s crucial to understand that this doesn’t diminish the significance of assessing both aspects in the clinical settings. Signs and symptoms offer valuable insights that complement each other, and it’s vital to weigh them together for an accurate diagnosis and effective management of DED. In these situations, differential diagnosis should also be considered including anterior blepharitis; allergic and infective conjunctivitis; eyelid abnormality or dysfunction leading to exposure (exposure keratopathy); nocturnal lagophthalmos (failure to close eyes at night).14
The diverse ways in which signs and symptoms present in different individuals highlight the complexity of DED. This underscores the importance of tailoring diagnostic and management approaches to each person’s unique needs and experiences.
Management & Advice
The 2025 TFOS DEWS III developed a prescribing algorithm enabling clinicians to develop a bespoke management regime tailored to address the etiological drivers contributing towards DED16.
It is crucial to understand that dry eye is unique to each patient. There’s no one-size-fits-all solution or magic drop that works for everyone. That’s why it’s essential to closely assess the patient, identify any factors that might be exacerbating their dry eye, and tailor the management plan accordingly.
Key factors from the experts16
- Experts outline a treatment approach based on the etiological drivers of the disease in an individual
- First line management includes ocular lifestyle modifications, tear film supplements and environmental adjustments to stabilise the tear film
- A REGIMEN comprising lubricants, lid hygiene, and the application of heat to the eyes for the treatment of dry eye disease and associated eye conditions.
- Preservatives such as benzalkonium chloride can result in toxic and pro-inflammatory effects, exacerbating dry eye signs and symptoms
- Omega-3s can play a critical role in improving clinical symptoms associated with inflammatory disorders of the ocular surface, including DED
Managing (DED) requires an individualised and comprehensive strategy that considers both the root causes and the unique characteristics of each patient. Artificial tears are a fundamental part of management and TFOS DEWS III highlights the value of improving meibomian gland health and making changes to daily habits.
The TFOS DEWS III prescribing algorithm offers a guide on how to manage patients dealing with dry eye based on the diagnostic tests performed to identify which of the etiological drivers are involved.16
You will see from the examples highlighted below that there is overlap with all 3 algorithms and the need for more than one intervention or management option may be required. (Topical lid hygiene and tear film supplementation/stabilisation feature in all algorithms acknowledging the importance of regimen of lubricants and lid hygiene (including heat) for the management of dry eye disease and associated conditions).
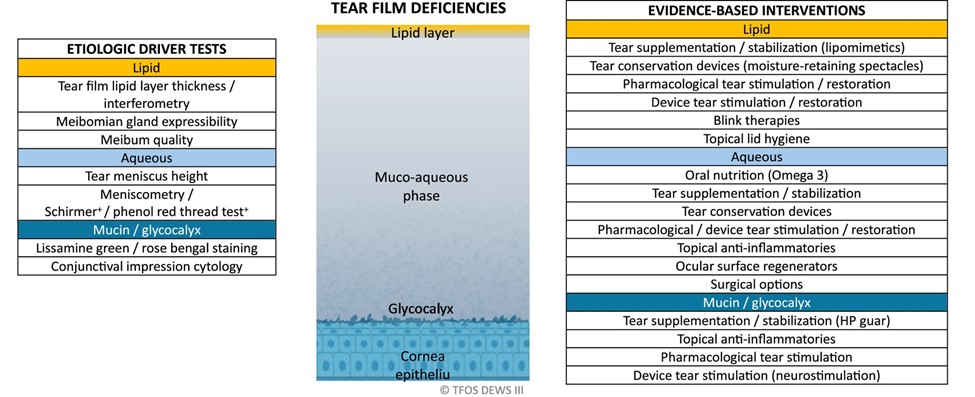
For example, if a lipid instability of the tear film is identified using the diagnostic criteria on the left-hand side, the table on the right-hand side suggests suitable evidence-based interventions such tear supplementation with a lipid component, and/or blink therapies, and/or topical lid hygiene as possible options.
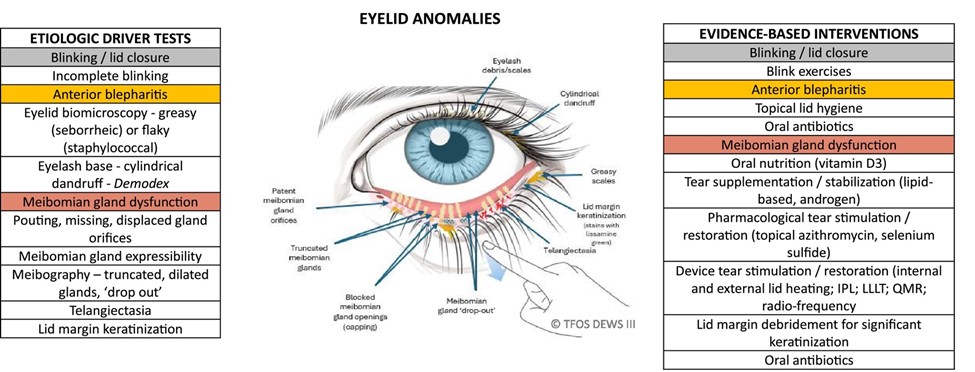
If MGD and or anterior blepharitis is identified, the Eyelid Anomalies algorithm suggests considering topical lid hygiene and/or tear film stabilisation and/or oral antibiotics as management recommendations.
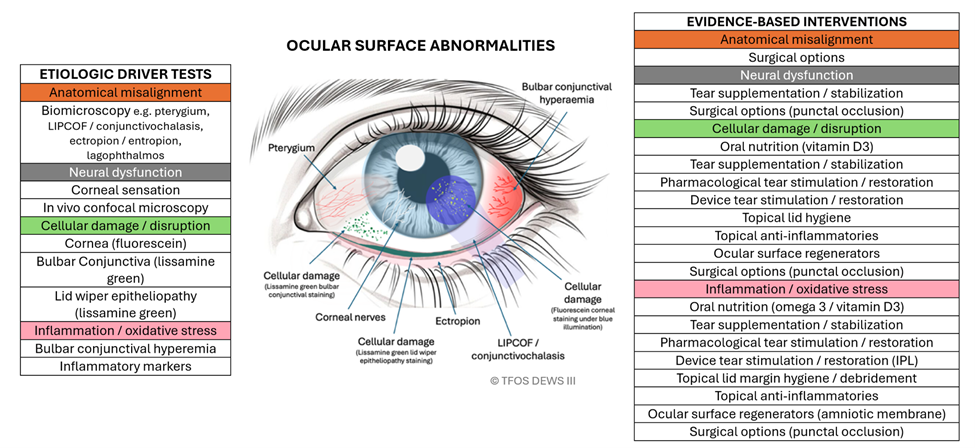
Corneal and/or conjunctival staining indicates an ocular surface abnormality, and this algorithm suggests tear film supplementation and/or topical lid hygiene and/or oral nutrition among the evidence-based interventions.
This article serves as an overview of the condition and treatment options. It does not serve as a clinical guidance. Eyecare provider guidelines should be used when managing patients.