Feeding the Retina: Nutrition and Age-Related Macular Degeneration
Dietary inflammation, micronutrient supplementation, and evolving insights into AMD prevention
Age-related macular degeneration (AMD) remains the leading cause of irreversible vision loss in adults over 50 worldwide. Its pathogenesis is multifactorial—genetic susceptibility, oxidative stress, chronic low-grade inflammation, and lifestyle all contribute—but nutrition is among the most modifiable. The landmark AREDS and AREDS2 trials established that targeted antioxidant supplementation can slow disease progression in selected patients1, and more recent evidence has broadened this view. Beyond isolated nutrients, overall diet quality and systemic inflammation are now recognized as powerful determinants of AMD risk and progression.
This article explores how nutrition influences AMD through multiple pathways: modulation of systemic inflammation, the role of specific micronutrients, mitochondrial health and resilience of the retinal pigment epithelium (RPE), and the emerging contribution of the gut microbiome.
Diet as a driver of inflammation
A 2025 systematic review and meta-analysis found that strong adherence to a Mediterranean diet- rich in fruits, vegetables, legumes, whole grains, fish, olive oil, and nuts, was linked to significantly lower risk and slower progression of AMD, with risk reductions of 34% in case-control studies and 23% in prospective cohorts.14 Beyond incidence, Mediterranean-style patterns were associated with slower geographic atrophy progression and a more gradual decline in visual acuity, particularly in non-central geographic atrophy stages. Protective elements include greater intake of vegetables and fruits alongside reduced red meat consumption.14
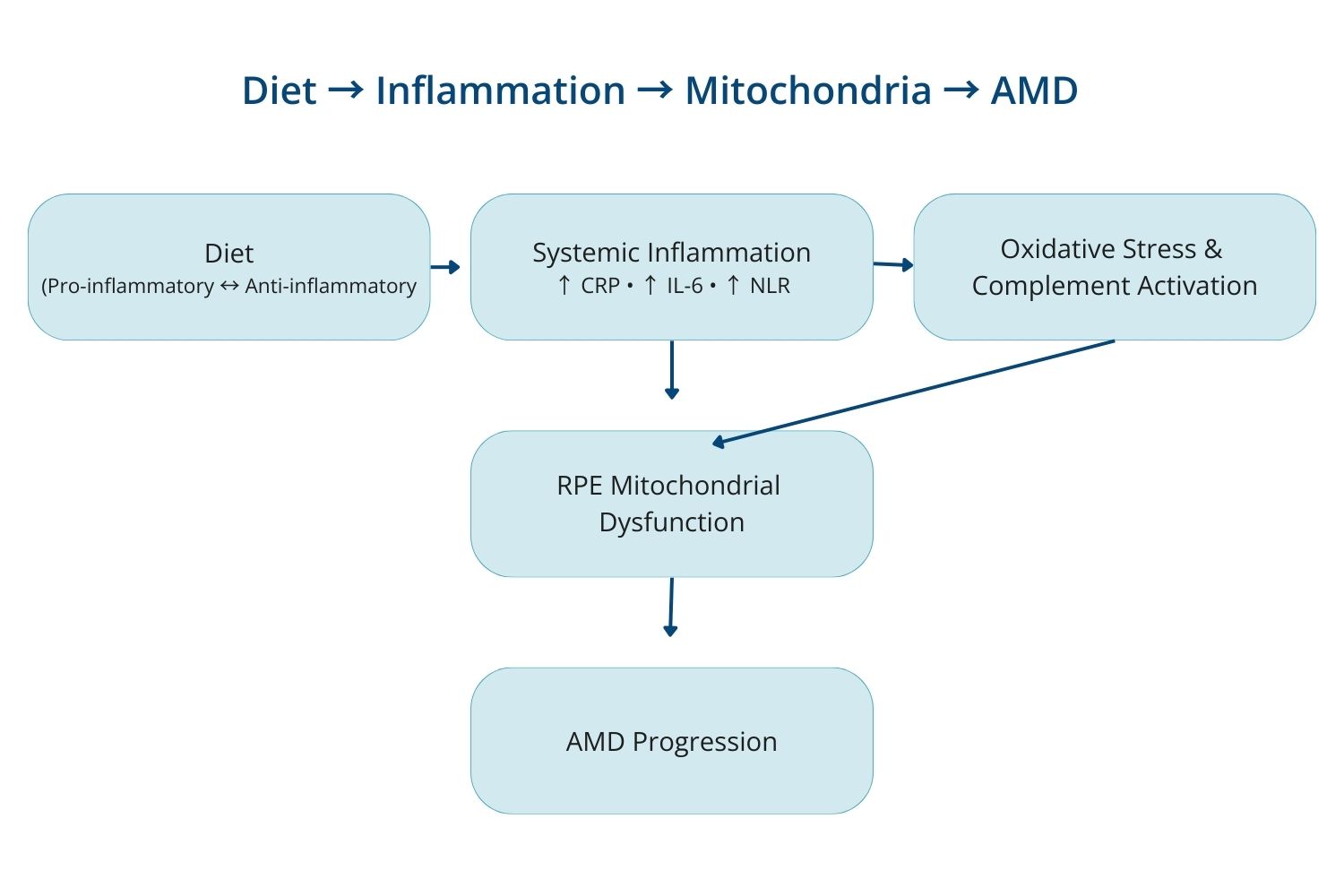
The biological rationale is clear. Diets high in refined carbohydrates, added sugars, red and processed meats, and trans fats fuel chronic low-grade inflammation through cytokine release, oxidative stress, and advanced glycation end-products. These patterns also drive systemic diseases like obesity and metabolic syndrome, which amplify inflammatory tone and accelerate AMD risk. By contrast, diets abundant in plant-based foods, fatty fish, and nuts provide phytonutrients, antioxidants, and omega-3 fatty acids that temper inflammation and support mitochondrial health. The Mediterranean diet epitomizes this balance, and its benefits now extend beyond cardiovascular protection to include reduced risk of AMD progression.2,4
Population-level data reinforce these observations. The Rotterdam Study showed that individuals with more pro-inflammatory diets, quantified by an adapted Dietary Inflammatory Index, faced significantly higher risk of AMD across early, intermediate, and late stages.2 Systemic inflammatory markers such as neutrophil-to-lymphocyte ratio (NLR), C-reactive protein (CRP), and interleukin-6 (IL-6) partly mediated this effect, underscoring systemic inflammation as the mechanistic bridge between poor diet and retinal degeneration.8,9
Mechanistic pathways
Systemic inflammation alters the retinal microenvironment through multiple mechanisms:
- Oxidative stress – pro-inflammatory diets elevate reactive oxygen species, damaging mitochondrial DNA and RPE integrity.
- Endothelial dysfunction – systemic inflammation impairs choroidal blood flow, reducing nutrient and oxygen delivery to the macula.
- Complement activation – dietary inflammation amplifies complement cascade activity, a well-established driver of AMD.
- Mitochondrial stress – persistent cytokine signaling accelerates mitochondrial dysfunction and promotes para-inflammation in the RPE.
Clinical implications
Mediterranean-style diets reduce systemic inflammatory load, improve biomarkers such as CRP and NLR, and support endothelial and mitochondrial health. Counseling AMD patients should extend beyond AREDS2 supplementation to include anti-inflammatory dietary guidance, steering patients toward Mediterranean-style eating patterns, encouraging omega-3–rich foods (e.g., fatty fish), and limiting processed sugars and fats.
Micronutrients and AMD
Carotenoids and antioxidants
The AREDS and AREDS2 trials remain the cornerstone of evidence, demonstrating that antioxidant supplementation can slow AMD progression in selected patients.1 Lutein and zeaxanthin, the dominant carotenoids concentrated in the macula, filter blue light, neutralize reactive oxygen species, and stabilize mitochondria in the retinal pigment epithelium (RPE). Higher dietary intake of these carotenoids is consistently associated with greater macular pigment optical density and lower risk of AMD progression. Other important antioxidants, vitamins C and E, both included in the AREDS2 formulation, help counter oxidative stress by neutralizing reactive oxygen species and reducing lipid peroxidation in retinal tissues, supporting the retina and retinal pigment epithelium against cumulative oxidative damage central to AMD pathogenesis.
A 2022 systematic review reaffirmed AREDS2 as the most evidence-based formulation. Strong support exists for lutein and zeaxanthin, while evidence for omega-3 fatty acids and flavonoids remains promising but inconclusive.1
Food sources: Dark leafy greens (spinach, kale, collard greens), corn, peas, broccoli, and egg yolks are particularly rich in lutein and zeaxanthin.
A recent randomized controlled trial found that eating 2 ounces of pistachios daily for 12 weeks significantly increased macular pigment optical density (MPOD) and serum lutein levels in adults with low baseline intake. Despite providing only modest lutein (∼1.6 mg/serving), pistachios doubled average U.S. intake and boosted MPOD to levels comparable with spinach or egg interventions, likely due to their lipid content enhancing absorption. Pistachios may therefore represent a simple, food-based strategy to strengthen retinal defenses in at-risk populations.15
While lutein and zeaxanthin are the primary carotenoids concentrated in the macula, astaxanthin has been explored as a complementary carotenoid that may further support retinal defense mechanisms.17 Its antioxidant and anti-inflammatory activity, and potential antiapoptotic effects, may help reduce oxidative stress and inflammatory signaling in retinal tissues, including the retinal pigment epithelium, which plays a central role in macular health.16,17 In the Carotenoids in Age-related Maculopathy Italian Study (CARMIS), a multi-nutrient formulation that included lutein/zeaxanthin and astaxanthin was associated with a greater likelihood of clinically meaningful stabilization or improvement in visual acuity, contrast sensitivity, and patient-reported visual function over 24 months compared with untreated subjects .16
B vitamins- especially folate, B6, and B12, have gained attention because elevated plasma homocysteine has been linked to AMD incidence and progression, likely through vascular dysfunction and oxidative stress.
In the Women’s Antioxidant and Folic Acid Cardiovascular Study (WAFACS), supplementation with folic acid (2.5 mg/day), vitamin B6 (50 mg/day), and vitamin B12 (1 mg/day) reduced the risk of developing AMD by 34% over 7 years in a large cohort of women at high cardiovascular risk.11Observational studies also suggest that inadequate intake of folate or B12 may accelerate retinal aging processes.
Food sources: Folate is abundant in legumes, leafy greens, citrus, and fortified grains. Vitamin B6 is found in poultry, fish, bananas, and potatoes. Vitamin B12 is obtained mainly from animal products such as dairy, eggs, meat, and fish. Patients on vegan diets should supplement Vitamin B12 to avoid deficiency.
Vitamin D
Vitamin D has strong biological plausibility in AMD through its anti-inflammatory, antioxidant, and anti-angiogenic properties. Experimental models demonstrate that vitamin D reduces oxidative stress and dampens inflammatory signaling in RPE and choroidal endothelial cells.
Observational studies often report lower 25(OH)D levels in AMD patients, though associations vary by population and subgroup. The VITAL-AMD randomized trial found that vitamin D₃ supplementation (2000 IU/day) did not reduce overall AMD incidence. However, participants with healthier baseline diets did experience protection, highlighting synergy between micronutrient intake and broader dietary patterns.3
Food sources: Fatty fish (salmon, mackerel, sardines), fortified dairy or plant milks, eggs, and sun exposure remain the primary sources of vitamin D.
Zinc and autophagy
Zinc is abundant in the retina, particularly in RPE melanosomes, where it supports antioxidant enzymes, mitochondrial stability, and proteostasis (protein homeostasis).
In AREDS/AREDS2, zinc supplementation slowed AMD progression in some patients1, though population data have been mixed. Mechanistically, zinc stimulates autophagy and mitophagy, clearing damaged proteins and lipofuscin; deficiency, by contrast, drives oxidative stress and impairs cellular clearance.10 Genetic variation in complement factor H (CFH) and ARMS2 may further modulate zinc’s effect, contributing to conflicting results in different populations.
Food sources: Oysters are the richest natural source of zinc, followed by other shellfish, red meat, poultry, beans, nuts, whole grains, and dairy.
Mitochondrial Health and AMD
The RPE is metabolically demanding, relying heavily on mitochondria to sustain photoreceptor function. With aging, and under stressors such as smoking, obesity, dyslipidemia, and poor diet- reactive oxygen species accumulate, damaging mitochondrial DNA and impairing ATP production.
In AMD, this is compounded by defective autophagy and mitophagy, leading to persistence of dysfunctional mitochondria, lipofuscin buildup, and chronic inflammation.
Two transcriptional hubs are central:
- NRF2 drives antioxidant enzyme production and cytoprotection.
- PGC-1α promotes mitochondrial biogenesis and efficient turnover of damaged organelles.
These pathways falter with aging, oxidative stress, and epigenetic changes, trapping the RPE in a vicious cycle: diminished NRF2/PGC-1α activity → impaired defenses and mitophagy → accumulation of dysfunctional mitochondria → more ROS and inflammation → accelerated degeneration.5,6
Therapeutic angles (preclinical):
- NRF2 activators (e.g., dimethyl fumarate; lutein/zeaxanthin cytoprotection).
- AMPK–PGC-1α activators to enhance mitochondrial biogenesis.
- Agents restoring autophagy/mitophagy.
Lifestyle interventions
Beyond diet, lifestyle factors such as regular physical activity, adequate sleep, and stress reduction support mitochondrial resilience by reducing oxidative load and stabilizing circadian mitochondrial rhythms. Incorporating these into patient counseling adds an accessible, non-pharmacologic layer to AMD management.
Practice pearl: Think beyond antioxidants- support the mitochondrial triad of defense, biogenesis, and clearance.
Photobiomodulation:
Photobiomodulation (PBM), or low-level light therapy, uses red to near-infrared wavelengths absorbed by cytochrome c oxidase in mitochondria, aiming to boost ATP production, reduce oxidative stress, and modulate inflammation, pathways central to AMD.
Early trials suggested modest benefits: a meta-analysis of three randomized studies (247 eyes) reported small gains in visual acuity and reduced drusen volume, though no impact on geographic atrophy.7 A more recent meta-analysis of six RCTs (360 patients) found no significant improvements in visual or structural outcomes compared with sham, with any short-term gains inconsistent and clinically negligible.13
Clinical perspective: PBM remains investigational. While it highlights the potential of targeting mitochondrial resilience in AMD, current evidence does not support routine use.
Proven approaches, dietary modification and AREDS2 supplementation,remain the foundation until larger, longer trials clarify PBM’s role.
Gut Microbiome Dysbiosis
Emerging evidence suggests that gut dysbiosis may also play a role in AMD pathogenesis. Animal models show that mice with choroidal neovascularization have significant shifts in gut microbiota compared to controls, and that high–glycemic diets worsen, while low–glycemic diets protect against disease, in part through microbial modulation. Mechanistically, dysbiosis favors pro-inflammatory bacterial taxa and reduces protective commensals, driving chronic low-grade inflammation, cytokine release, and disruption of the blood–retinal barrier. In humans, findings remain preliminary but point to a reduced Firmicutes/Bacteroidetes ratio and altered metabolic pathways, including glutamate degradation and fatty acid elongation in AMD patients, suggesting microbial and metabolic signatures linked to retinal dysfunction. While clinical studies are still limited, these data highlight the gut–retina axis as a promising target, and support dietary strategies such as Mediterranean or low-glycemic eating patterns to promote eubiosis and reduce systemic inflammatory load.12
How Can Clinicians Integrate Evidence-Based Diet and Lifestyle Advice into Their Routine Practice?
Nutrition is central to AMD prevention and management. Dietary interventions are most impactful when adopted early, but they remain valuable after diagnosis in slowing progression and supporting retinal resilience. Advances in genetics and metabolomics will continue to refine our ability to personalize nutritional guidance, helping identify which patients benefit most from targeted strategies.
AMD care has entered a phase where diet quality is not optional background advice—it is a modifiable risk factor. Clinicians can integrate this without adding burden by using a simple, repeatable workflow:
- Screen for dietary habits with targeted questions:
“How often do you eat vegetables?” “How often do you eat fish?” “How often do you eat sweets/white bread/processed snacks?”
Assessing intake quickly reveals actionable pathways. In 2019, only 12.3% and 10.0% of U.S. adults met fruit and vegetable intake recommendations, respectively, highlighting a major opportunity for intervention. - Provide clear anti-inflammatory targets:
A whole-food, anti-inflammatory pattern with low–glycemic swaps and regular omega-3–rich foods. - Layer supplements appropriately:
Use supplementation when indicated, correct deficiencies, and consider targeted nutrient testing. - Anchor the plan with one behavior:
Choose one dietary change plus one lifestyle support (sleep, physical activity, or stress regulation). - Track and reinforce:
Review a short 3–5-day food and beverage log at follow-up, identify patterns, and adjust with 1–2 concrete next steps.
Keep current: Seek out reputable, evidence-based CE and professional networks in ocular wellness and nutrition, such as scope-connect.com and the Ocular Wellness & Nutrition Society, to ensure your recommendations stay aligned with the latest research.
Final Takeaway
The most powerful nutrition message for AMD is not a single nutrient, it is a pattern. Consistent, whole-food dietary habits lower systemic inflammation and support the metabolic demands of the macula. By combining targeted supplementation when appropriate, correcting deficiencies, and counseling patients on diet and lifestyle strategies that promote mitochondrial resilience and systemic health, clinicians can offer a practical, holistic pathway to preserving vision across the course of AMD.
References
- Pameijer EM, Heus P, Damen JAA, et al. What did we learn in 35 years of research on nutrition and supplements for age-related macular degeneration: a systematic review. Acta Ophthalmol. 2022;100(8):e1541–e1552.
- Vergroesen JE, Thee EF, de Crom TOE, et al. The inflammatory potential of diet is associated with the risk of age-related eye diseases. Clin Nutr. 2023;42(12):2404–2413. doi:10.1016/j.clnu.2023.10.008.
- Parekh N, Albert CM, Buring JE, et al. Effect of vitamin D3 on risk of age-related macular degeneration in the VITAL randomized clinical trial. 2024;331(4):341–349.
- Han X, Lains I, Li J, et al. Integrating genetics and metabolomics from multi-ethnic and multi-fluid data reveals putative mechanisms for age-related macular degeneration. Cell Rep Med. 2023;4(7):101085. doi:10.1016/j.xcrm.2023.101085.
- Kaarniranta K, Uusitalo H, Blasiak J, et al. Mechanisms of mitochondrial dysfunction and their impact on age-related macular degeneration. Prog Retin Eye Res. 2020;79:100858.
- Mehrzadi S, Hemati K, Reiter RJ, Hosseinzadeh A. Mitochondrial dysfunction in age-related macular degeneration: melatonin as a potential treatment. Expert Opin Ther Targets. 2020;24(4):359–378.
- Rassi TNO, Barbosa LM, Pereira S, et al. Photobiomodulation efficacy in age-related macular degeneration: a systematic review and meta-analysis of randomized clinical trials. Int J Retina Vitreous. 2024;10(1):54.
- Seddon JM, Gensler G, Milton RC, Klein ML, Rifai N. Association between C-reactive protein and age-related macular degeneration. 2004;291(6):704–710.
- Seddon JM, George S, Rosner B, Rifai N. Progression of age-related macular degeneration: prospective assessment of C-reactive protein, interleukin 6, and other cardiovascular biomarkers. Arch Ophthalmol. 2005;123(6):774–782.
- Blasiak J, Pawlowska E, Chojnacki J, Szczepanska J, Chojnacki C, Kaarniranta K. Zinc and autophagy in age-related macular degeneration. Int J Mol Sci. 2020;21(14):4994.
- Chan HN, Zhang XJ, Ling XT, et al. Vitamin D and ocular diseases: a systematic review. Int J Mol Sci. 2022;23(8):4226.
- Zysset-Burri DC, Keller I, Berger LE, Largiadèr CR, Wittwer M, Wolf S, et al. Associations of the intestinal microbiome with the complement system in neovascular age-related macular degeneration. NPJ Genom Med. 2020;5:34.
- Chen KY, Lee HK, Chan HC, Chan CM. Is multiwavelength photobiomodulation effective and safe for age-related macular degeneration? A systematic review and meta-analysis. Ophthalmol Ther. 2025;14(5):969–987.
- Agrón E, Vance E, Domalpally A, Chew EY, Keenan TDL. Relationships between diet and geographic atrophy progression in the Age-Related Eye Diseases Studies 1 and 2. 2025;17(5):771.
- Scott TM, Ogunbodede O, McKay DL, Johnson EJ. Pistachio consumption increases macular pigment optical density in healthy adults: a randomized controlled trial. J Nutr. 2025;155(1):168–174.
- Piermarocchi, S., Saviano, S., Parisi, V., Tedeschi, M., Panozzo, G., Scarpa, G., Boschi, G., Lo Giudice, G. & Carmis Study Group (2012) ‘Carotenoids in Age-related Maculopathy Italian Study (CARMIS): two-year results of a randomized study’, European Journal of Ophthalmology, 22(2), pp. 216-225. doi:10.5301/ejo.5000069
- Giannaccare, G., Pellegrini, M., Senni, C., Bernabei, F., Scorcia, V. & Cicero, A.F.G. (2020) ‘Clinical Applications of Astaxanthin in the Treatment of Ocular Diseases: Emerging Insights’, Marine Drugs, 18(5), p. 239. doi: 10.3390/md18050239.
- Centers for Disease Control and Prevention (2022) Adults meeting fruit and vegetable intake recommendations — United States, 2019. MMWR Morbidity and Mortality Weekly Report, 71(1), pp. 1-9. Available at: https://www.cdc.gov/mmwr/volumes/71/wr/mm7101a1.htm (Accessed: 6 November 2025).
Latest Articles

HCP Popup
Are you a healthcare or eye care professional?
The information contained on this website is provided exclusively for healthcare and eye care professionals and is not intended for patients.
Click ‘Yes’ below to confirm that you are a healthcare professional and agree to the terms of use.
If you select ‘No’, you will be redirected to scopeeyecare.com
This will close in 0 seconds
